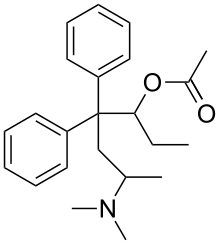
Acetylmethadol
 | |
| Clinical data | |
|---|---|
| ATC code | None |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | 509-74-0 |
| PubChem (CID) | 10517 |
| DrugBank | DB01433 |
| ChemSpider | 10080 |
| Chemical and physical data | |
| Formula | C23H31NO2 |
| Molar mass | 353.498 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
Acetylmethadol (INN), also known as methadyl acetate (USAN), is a synthetic opioid analgesic.[2] It is a racemic mixture of alphacetylmethadol (α-acetylmethadol) and betacetylmethadol (β-acetylmethadol), which are in turn racemic mixtures of levacetylmethadol (LAAM; L-α-acetylmethadol) and D-α-acetylmethadol and L-β-acetylmethadol and D-β-acetylmethadol, respectively.[2] Hence, acetylmethadol has four possible optical isomers.[2] All of these isomers have been shown to partially or fully substitute for the discriminative stimulus effects of heroin in rats, and thus it can be inferred that, in addition to LAAM which is used clinically as such, they are all likely to be active opioid analgesics in humans.[2]
In the United States, acetylmethadol and its individual isomers are all Schedule I drugs under the Controlled Substances Act, except LAAM, which is Schedule II and was used clinically until 1993. Acetylmethadol has an ACSCN of 9601. The isomers have individual ACSCNs, which are as follows: Alphacetylmethadol 9603, Betacetylmethadol 9607, LAAM 9648. All of the above have annual manufacturing quotas of 2 grammes as of 2013, except LAAM, which is 4 grammes.[3]
References
- ↑ Richard Lawrence Miller (30 December 2002). The Encyclopedia of Addictive Drugs. Greenwood Publishing Group. p. 222. ISBN 978-0-313-31807-8. Retrieved 15 May 2012.
- 1 2 3 4 Newman JL, Vann RE, May EL, Beardsley PM (October 2002). "Heroin discriminative stimulus effects of methadone, LAAM and other isomers of acetylmethadol in rats". Psychopharmacology. 164 (1): 108–14. doi:10.1007/s00213-002-1198-8. PMID 12373424.
- ↑ 21 U.S.C. § 812(b)(1)
| Opioids | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Paracetamol-type |
| ||||||||||||||||||||||||||||||
| NSAIDs |
| ||||||||||||||||||||||||||||||
| Cannabinoids | |||||||||||||||||||||||||||||||
| Ion channel modulators |
| ||||||||||||||||||||||||||||||
| Myorelaxants | |||||||||||||||||||||||||||||||
| Others | |||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||
| MOR |
|
|---|---|
| DOR |
|
| KOR |
|
| NOP |
|
| Unsorted |
|
| Others |
|
See also: Peptide receptor modulators | |