Rabaptin
| Rabaptin | |||||||||
|---|---|---|---|---|---|---|---|---|---|
|
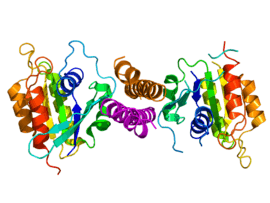
Crystallographic structure of the C-terminal Rabaptin5 domain homodimer (colored orange and magenta at the center of the figure) complexed with two independent molecules of the GTPase domain of RAB5A (rainbow colored, N-terminus = blue, C-terminus = red).[1] | |||||||||
| Identifiers | |||||||||
| Symbol | Rabaptin | ||||||||
| Pfam | PF03528 | ||||||||
| InterPro | IPR003914 | ||||||||
| |||||||||
Rabaptin is a key protein involved in regeneration of injured axons.
Regeneration of injured axons at neuromuscular junctions is regulated by extra-cellular protein factors that promote neurite outgrowth. A novel neurite outgrowth factor from chick denervated skeletal muscle has been cloned and characterised. The protein, termed neurocrescin (rabaptin), has been shown to be secreted in an activity-dependent fashion.[2]
Rabaptin is a 100kDa coiled-coil protein that interacts with the GTP form of the small GTPase Rab5 (see RAB5A, RAB5B, RAB5C) a potent regulator of endocytic transport.[3] It is mainly cytosolic, but a fraction co-localises with Rab5 to early endosomes. Rab5 recruits rabaptin-5 to purified early endosomes in a GTP-dependent manner, demonstrating functional similarities with other members of the Ras family. Immunodepletion of rabaptin-5 from cytosol strongly inhibits Rab5-dependent early endosome membrane fusion. Thus, rabaptin-5 is a Rab effector required for membrane docking and fusion.
Rab5 contributes to early endosome fusion that works through the coordination of other effector proteins, as mentioned above. Rabaptin components may serve to control the SNARE activity which are vital in membrane fusion.[4]
Structure
A crystal structure of the GTPase domain of RAB5A complexed with the C-terminal domain of Rabaptin5 has been determined.[1] The two proteins form a symmetric RAB5A-Rabaptin5 ternary complex with a parallel coiled-coil Rabaptin5 homodimer in the middle complexed with two molecules of Rabaptin5, one on each side (see figure to the right).
Human proteins
RABEP1; RABEP2;
References
- 1 2 PDB: 1tu3; Zhu G, Zhai P, Liu J, Terzyan S, Li G, Zhang XC (October 2004). "Structural basis of Rab5-Rabaptin5 interaction in endocytosis". Nat. Struct. Mol. Biol. 11 (10): 975–83. doi:10.1038/nsmb832. PMID 15378032.
- ↑ Nishimune H, Uyeda A, Nogawa M, Fujimori K, Taguchi T (1997). "Neurocrescin: a novel neurite-outgrowth factor secreted by muscle after denervation". NeuroReport. 8 (16): 3649–3654. doi:10.1097/00001756-199711100-00045. PMID 9427343.
- ↑ Zerial M, Stenmark H, Vitale G, Ullrich O (1995). "Rabaptin-5 is a direct effector of the small GTPase Rab5 in endocytic membrane fusion". Cell. 83 (3): 423–432. doi:10.1016/0092-8674(95)90120-5. PMID 8521472.
- ↑ Deneka M, Neeft M, Popa I, van Oort M, Sprong H, Oorschot V, Klumperman J, Schu P, van der Sluijs P (June 2003). "Rabaptin-5alpha/rabaptin-4 serves as a linker between rab4 and gamma(1)-adaptin in membrane recycling from endosomes". EMBO J. 22 (11): 2645–57. doi:10.1093/emboj/cdg257. PMC 156754
 . PMID 12773381.
. PMID 12773381.
This article incorporates text from the public domain Pfam and InterPro IPR003914