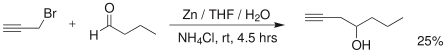Propargyl bromide
 | |
| Names | |
|---|---|
| Preferred IUPAC name
3-Bromoprop-1-yne | |
| Other names
3-Bromo-1-propyne Bromopropyne 1-Brom-2-propin 1-Bromo-2-propyne Gamma-bromoallylene 1-Bromo-2-propyne 2-Propynyl bromide Propargyl bromide Propynyl bromide gamma-Bromoallylene | |
| Identifiers | |
| 106-96-7 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 7554 |
| ECHA InfoCard | 100.003.135 |
| PubChem | 7842 |
| UNII | F3H7ZXK9ZU |
| |
| |
| Properties | |
| C3H3Br | |
| Molar mass | 118.96 g·mol−1 |
| Appearance | colourless liquid |
| Density | 1.57 g/mL (20 °C)[1] |
| Melting point | −61.1 °C (−78.0 °F; 212.1 K)[1] |
| Boiling point | 89 °C (192 °F; 362 K)[1] |
| Insoluble | |
| Solubility | Soluble in organic solvents |
| log P | 1.179 |
| Vapor pressure | 72 mbar (20 °C)[1] |
| Hazards | |
| Main hazards | Highly Flammable, Toxic, Corrosive |
| NFPA 704 | |
| Flash point | 18 °C (64 °F; 291 K)[1] |
| 324 °C (615 °F; 597 K)[1] | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Propargyl bromide, also known as 3-bromo-1-propyne, is an organic compound with the chemical formula CHCCH2Br. It is a halogenated organic compound consisting of an alkyl functional group with 2-propynyl group linked to bromide. It has a lachrymatory effect, like related compounds. The compound is a useful reagent in organic synthesis.
Applications
In the 1960s, propargyl bromide was first used in a soil fumigant called Trizone.[2]
Propargyl bromide can also be used as an intermediate for the synthesis of organic compounds, including agrochemicals and pharmaceuticals. It forms a Grignard reagent at low temperatures, for example.[3]
Production
Propargyl bromide may be produced by the treatment of propargyl alcohol with phosphorus tribromide:[4]
Reactions
Propargyl bromide can be used in enyne metathesis of propargylic amines, propargylation of spiro ketones, production of allylic alcohols, and enone complexes.[5]
Aldehydes may be reacted with propargyl bromide in a Barbier-type reaction to yield alkyne alcohols as well:[6]
Safety
Propargyl bromide is a lachrymator and an alkylating agent.[7]
See also
References
- 1 2 3 4 5 6 Record in the GESTIS Substance Database of the IFA
- ↑ Franz Müller and Arnold P. Applebyki "Weed Control, 2. Individual Herbicides" in Ullmann's Encyclopedia of Industrial Chemistry 2010 doi:10.1002/14356007.o28_o01
- ↑ Henning Hopf, Ingrid Böhm, and Jürgen Kleinschroth (1990). "Diels-Alder Reaction of 1,2,4,5-Hexatetraene: Tetramethyl[2.2]paracyclophane-4,5,12,13-tetracarboxylate". Org. Synth. 60: 41.; Coll. Vol., 7, p. 485
- ↑ "Process for Producing Propargyl Bromide". Retrieved November 7, 2012.
- ↑ "Propargyl Bromide". Retrieved November 5, 2012.
- ↑ Artur Jõgi & Uno Mäeorg (2001). "Zn Mediated Regioselective Barbier Reaction of Propargylic Bromides in THF/aq. NH4Cl Solution" (PDF). Molecules. 6 (12): 964–968. doi:10.3390/61200964. ISSN 1420-3049.
- ↑ "3-Bromo-1-Propyne". Retrieved November 3, 2012.