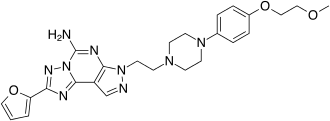
Preladenant
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code | none |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
377727-87-2 |
| PubChem (CID) | 10117987 |
| IUPHAR/BPS | 5614 |
| ChemSpider |
8293510 |
| UNII |
950O97NUPO |
| ECHA InfoCard | 100.210.813 |
| Chemical and physical data | |
| Formula | C25H29N9O3 |
| Molar mass | 503.555 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Preladenant (SCH 420814) was a drug that was developed by Schering-Plough which acted as a potent and selective antagonist at the adenosine A2A receptor. It was being researched as a potential treatment for Parkinson's disease.[1] Positive results were reported in Phase II clinical trials in humans,[2] but it did not prove itself to be more effective than a placebo during Phase III trials, and so was discontinued in May 2013.[3]
References
- ↑ Hodgson RA, Bertorelli R, Varty GB, Lachowicz JE, Forlani A, Fredduzzi S, Cohen-Williams ME, Higgins GA, Impagnatiello F, Nicolussi E, Parra LE, Foster C, Zhai Y, Neustadt BR, Stamford AW, Parker EM, Reggiani A, Hunter JC (March 2009). "Characterization of the Potent and Highly Selective A2A Receptor Antagonists Preladenant and SCH 412348 in Rodent Models of Movement Disorders and Depression". The Journal of Pharmacology and Experimental Therapeutics. 330 (1): 294–303. doi:10.1124/jpet.108.149617. PMID 19332567.
- ↑ Hauser, R. A.; Cantillon, M.; Pourcher, E.; Micheli, F.; Mok, V.; Onofrj, M.; Huyck, S.; Wolski, K. (2011). "Preladenant in patients with Parkinson's disease and motor fluctuations: a phase 2, double-blind, randomised trial". The Lancet Neurology. 10 (3): 221–229. doi:10.1016/S1474-4422(11)70012-6.
- ↑ http://bigstory.ap.org/article/merck-ends-development-parkinsons-disease-drug
This article is issued from Wikipedia - version of the 9/5/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.