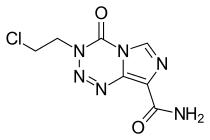
Mitozolomide
 | |
| Clinical data | |
|---|---|
| ATC code | none |
| Identifiers | |
| |
| CAS Number |
85622-95-3 |
| PubChem (CID) | 71766 |
| ChemSpider |
64805 |
| UNII |
E3U7286V3W |
| ChEMBL |
CHEMBL435951 |
| ECHA InfoCard | 100.079.921 |
| Chemical and physical data | |
| Formula | C7H7ClN6O2 |
| Molar mass | 242.622 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Mitozolomide (INN) is an antineoplastic. It is an imidazotetrazine derivative.
Development of mitozolomide was discontinued during Phase II clinical trials after it was found to cause severe and unpredictable bone marrow suppression.[1] Temozolomide, which has been in clinical use since 1999, is a less toxic analogue of mitozolomide.[2]
References
- ↑ Fairbairn LJ, Chinnasamy N, Lashford LS, Chinnasamy D, Rafferty JA (February 2000). "Enhancing hemopoietic drug resistance: a rationale for reconsidering the clinical use of mitozolomide" (PDF). Cancer Gene Ther. 7 (2): 233–9. doi:10.1038/sj.cgt.7700120. PMID 10770631.
- ↑ Newlands ES, Blackledge GR, Slack JA, et al. (February 1992). "Phase I trial of temozolomide (CCRG 81045: M&B 39831: NSC 362856)". Br J Cancer. 65 (2): 287–91. doi:10.1038/bjc.1992.57. PMC 1977719
 . PMID 1739631.
. PMID 1739631.
This article is issued from Wikipedia - version of the 8/30/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.