Herd immunity

Herd immunity (also called herd effect, community immunity, population immunity, or social immunity) is a form of indirect protection from infectious disease that occurs when a large percentage of a population has become immune to an infection, thereby providing a measure of protection for individuals who are not immune.[1][2] In a population in which a large number of individuals are immune, chains of infection are likely to be disrupted, which stops or slows the spread of disease.[3] The greater the proportion of individuals in a community who are immune, the smaller the probability that those who are not immune will come into contact with an infectious individual.[1]
Individual immunity can be gained through recovering from a natural infection or through artificial means such as vaccination.[3] Some individuals cannot become immune due to medical reasons and in this group herd immunity is an important method of protection.[4][5] Once a certain threshold has been reached, herd immunity gradually eliminates a disease from a population.[5] This elimination, if achieved worldwide, may result in the permanent reduction in the number of infections to zero, called eradication.[6] This method was used for the eradication of smallpox in 1977 and for the regional elimination of other diseases.[7] Herd immunity does not apply to all diseases, just those that are contagious, meaning that they can be transmitted from one individual to another.[5] Tetanus, for example, is infectious but not contagious, so herd immunity does not apply.[4]
The term herd immunity was first used in 1923.[1] It was recognized as a naturally occurring phenomenon in the 1930s when it was observed that after a significant number of children had become immune to measles, the number of new infections temporarily decreased, including among susceptible children.[8] Mass vaccination to induce herd immunity has since become common and proved successful in preventing the spread of many infectious diseases.[9] Opposition to vaccination has posed a challenge to herd immunity, allowing preventable diseases to persist in or return to communities that have inadequate vaccination rates.[10][11][12]
Effects
Protection of those without immunity

Some individuals depend on herd immunity because they either cannot develop immunity or for medical reasons cannot be vaccinated.[4][14] Newborn infants are too young to receive many vaccines, either for safety reasons or because passive immunity renders the vaccine ineffective.[15] Individuals who possess an immunodeficiency from HIV/AIDS, lymphoma, leukemia, a bone marrow cancer, an impaired spleen, chemotherapy, or radiotherapy may have lost any immunity that they previously had and vaccines may not be of any use for them because of their immunodeficiency.[4][14][15][16] Vaccines are typically imperfect as some individuals' immune systems may not generate an adequate immune response to vaccines to confer long-term immunity, so a portion of those who are vaccinated may lack immunity.[1] Lastly, vaccine contraindications for specific populations may prevent individuals in these populations from becoming immune.[14][17][18][19] Individuals in one of these groups may be at a greater risk of developing complications from infection because of their medical status, so they generally rely on herd immunity more than others who can otherwise safely become immune via vaccination.[4][14][19][20]
High levels of immunity in one age group can create herd immunity for other age groups.[7] Vaccinating adults against pertussis reduces pertussis incidence in infants too young to be vaccinated, who are at the greatest risk of complications from the disease.[21][22] This is especially important for close family members, who account for most of the transmissions to young infants.[7][19] In the same manner, children receiving vaccines against pneumococcus reduces pneumococcal disease incidence among younger, unvaccinated siblings.[23] Conversely, vaccinating children against pneumococcus and rotavirus has had the effect of reducing pneumococcus- and rotavirus-attributable hospitalizations for older children and adults, who do not normally receive these vaccines.[23][24][25] Influenza (flu) is more severe in the elderly than in younger age age groups, but influenza vaccines lack effectiveness in this demographic due to a waning of the immune system with age.[7][26] The prioritization of school-age children for seasonal flu immunization, which is more effective than vaccinating the elderly, however, has shown to create a certain degree of herd immunity for the elderly.[7][26]
For sexually transmitted infections (STIs), high levels of immunity in one sex induces herd immunity for both sexes.[9][27][28] Vaccines against STIs that are targeted at one sex result in significant declines in sexual disease in both sexes if vaccine uptake in the target sex is high.[27][28][29] Herd immunity from female vaccination does not, however, extend to homosexual males.[28] If vaccine uptake among the target sex is low, then the other sex may need to be immunized so that that sex can be sufficiently protected.[27][28] High-risk behaviors make eliminating STIs difficult since even though most infections occur among individuals with moderate risk, the majority of transmissions occur because of individuals who engage in high-risk behaviors.[9] For these reasons, certain populations may require immunizing high-risk persons or individuals of both sexes to establish herd immunity.[9][28]
Evolutionary pressure
Herd immunity itself acts as an evolutionary pressure on certain viruses, influencing viral evolution by encouraging the production of novel strains, in this case referred to as escape mutants, that are able to "escape" from herd immunity and spread more easily.[30][31] At the molecular level, viruses escape from herd immunity through antigenic drift, which is when mutations accumulate in the portion of the viral genome that encodes for the virus's surface antigen, typically a protein of the virus capsid, producing a change in the viral epitope.[32][33] Alternatively, the reassortment of separate viral genome segments, or antigenic shift, which is more common when there are more strains in circulation, can also produce new serotypes.[30][34] When either of these occur, memory T cells no longer recognize the virus, so herd immunity ceases to be relevant to the dominant circulating strain.[33][34] For both influenza and norovirus, epidemics temporarily induce herd immunity until a new dominant strain emerges, causing successive waves of epidemics.[32][34] As this evolution poses a challenge to herd immunity, broadly neutralizing antibodies and "universal" vaccines that can provide protection beyond a specific serotype are in development.[31][35][36]
Serotype replacement
Serotype replacement, or serotype shifting, may occur if the prevalence of a specific serotype declines due to high levels of immunity, allowing other serotypes to replace it.[37][38] Initial vaccines against Streptococcus pneumoniae significantly reduced nasopharyngeal carriage of vaccine serotypes (VTs), including antibiotic-resistant types,[23][39] only to be entirely offset by increased carriage of non-vaccine serotypes (NVTs).[23][37][38] This did not result in a proportionate increase in disease incidence though since NVTs were less invasive than VTs.[37] Since then, pneumococcal vaccines that provide protection from the emerging serotypes have been introduced and have successfully countered their emergence.[23] The possibility of future shifting remains, so further strategies to deal with this include expansion of VT coverage and the development of vaccines that use either killed whole-cells, which have more surface antigens, or proteins present in multiple serotypes.[23][40]
Eradication of diseases

If herd immunity has been established and maintained in a population for a sufficient time, the disease is inevitably eliminated—no more endemic transmissions occurs.[5] If elimination is achieved worldwide and the number of cases is permanently reduced to zero, then a disease can be declared eradicated.[6] Eradication can thus be considered the final effect or end-result of public health initiatives to control the spread of infectious disease.[6][7] The benefits of eradication include ending all morbidity and mortality caused by the disease, financial savings for individuals, health care providers, and governments, and enabling resources used to control the disease to be used elsewhere.[6] To date, two diseases have been eradicated using herd immunity and vaccination: rinderpest and smallpox.[1][7][41] Eradication efforts that rely on herd immunity are currently underway for poliomyelitis, though civil unrest and distrust of modern medicine have made this difficult.[1][42] Voluntary vaccination, for a variety of reasons, may be an obstacle to eradication if not enough people become immune to a disease.[43][44][45][46]
Free riding
Herd immunity is vulnerable to the free rider problem.[47] Individuals who lack immunity, primarily those who choose not to vaccinate, free ride off the herd immunity created by those who are immune.[47] As the number of free riders in a population increases, outbreaks of preventable diseases become more common and more severe.[10][11][12] Individuals may choose to free ride for a variety of reasons, including bandwagoning or groupthinking,[44][48] social norms or peer pressure,[49] religious beliefs,[11] the perceived effectiveness of a vaccine,[49] mistrust of vaccines or public health officials,[50] and believing that the risks associated with vaccines are greater than those associated with infection.[1][11][12][49] Individuals are more likely to free ride if vaccination rates are high enough so as to convince a person that he or she may not need to be immune since a sufficient number of others already are.[1][46] If large numbers of people in a community free ride, herd immunity in that community is lost.[44][46]
Mechanism
| Disease | Transmission | R0 | HIT |
|---|---|---|---|
| Measles | Airborne | 12–18 | 92–95% |
| Pertussis | Airborne droplet | 12–17[52] | 92–94% |
| Diphtheria | Saliva | 6–7 | 83–86% |
| Rubella | Airborne droplet | ||
| Smallpox | 5–7 | 80–86% | |
| Polio | Fecal-oral route | ||
| Mumps | Airborne droplet | 4–7 | 75–86% |
| SARS | 2–5[53] | 50–80% | |
| Ebola (Ebola virus epidemic in West Africa) | Bodily fluids | 1.5–2.5[54] | 33–60% |
| Influenza (influenza pandemics) | Airborne droplet | 1.5–1.8[52] | 33–44% |
Individuals who are immune to a disease act as a barrier in the spread of disease, slowing or preventing the transmission of disease to others.[3] An individual's immunity can be acquired via a natural infection or through artificial means, such as vaccination.[3] When a critical proportion of the population becomes immune, called the herd immunity threshold (HIT) or herd immunity level (HIL), the disease may no longer persist in the population, ceasing to be endemic.[5][30] This threshold can be calculated by taking R0, the basic reproduction number, or the average number of new infections caused by each case in an entirely susceptible population that is homogeneous, or well-mixed, meaning each individual can come into contact with every other susceptible individual in the population,[9][30][43] and multiplying it by S, the proportion of the population who are susceptible to infection:
S can be rewritten as (1 - p) because p is the proportion of the population that is immune and p + S equals one. Then, the equation can be rearranged to place p by itself as follows:
- → →
With p being by itself on the left side of the equation, it can now be written as pc to represent the critical proportion of the population needed to become immune to stop the transmission of disease, or the herd immunity threshold.[9] R0 functions as a measure of contagiousness, so low R0 values are associated with lower HITs needed, whereas higher R0s demand higher HITs.[30][43] For example, a disease with an R0 of 2 theoretically needs only a 50% HIT to be eliminated, whereas a disease with an R0 of 10 needs a 90% HIT.[30] These calculations assume that the entire population is susceptible, meaning no individuals are immune to the disease. In reality, varying proportions of the population are immune to any given disease at any given time.[9] To account for this, the effective reproductive number Re, also written as Rt, or the average number of infections caused at time t, can found by multiplying R0 by the fraction of the population that is still susceptible. When Re is reduced to and sustained below 1, the number of cases occurring in the population gradually decreases until the disease has been eliminated.[9][30][55] If a population is immune to a disease in excess of that disease's HIT, the number of cases reduces at a faster rate, outbreaks are even less likely to happen, and outbreaks that occur are smaller than they would be otherwise .[1][9] If Re increases to above 1, then the disease is neither in a steady state nor decreasing in incidence but is actively spreading through the population and infecting a larger number of people than usual.[44][55]
A second assumption in these calculations is that populations are homogeneous, or well-mixed, meaning that every individual comes into contact with every other individual, when in reality populations are better described as social networks as individuals tend to cluster together, remaining in relatively close contact with a limited number of other individuals. In these networks, transmission only occurs between those who are geographically or physically close to one another.[1][43][44] The shape and size of a network is likely to alter a disease's HIT, making incidence either more or less common.[30][43] In heterogeneous populations, R0 is now considered to be a measure of the number of cases generated by a "typical" infectious person, which depends on how individuals within a network interact with each other.[1] Interactions within networks are more common than between networks, in which case the most highly connected networks transmit disease more easily, resulting in a higher R0 and a higher HIT than would be required in a less connected network.[1][44] In networks that either opt not to become immune or are not immunized sufficiently, diseases may persist despite not existing in better-immunized networks.[44]
Boosts
Vaccination
The primary way to boost levels of immunity in a population is through vaccination.[1][56] Their use is originally based on the observation that milkmaids exposed to cowpox were immune to smallpox, so the practice of inoculating people with the cowpox virus began as a way to prevent smallpox cases.[42] Well-developed vaccines provide protection in a far safer way than natural infections, as vaccines generally do not cause the diseases they protect against and severe adverse effects are significantly less common than complications from natural infections.[57][58] The immune system does not distinguish between natural infections and vaccines, forming an active response to both, so immunity induced via vaccination is similar to what would have occurred from contracting and recovering from the disease.[59] To achieve herd immunity through vaccination, vaccine manufacturers aim to produce vaccines with low failure rates and policy makers aim to encourage their use.[56] After the successful introduction and widespread use of a vaccine, sharp declines in the incidence of diseases it protects against can be observed, necessarily decreasing the number of hospitalizations and deaths caused by such diseases.[60][61][62]
Assuming a vaccine is 100% effective, then the equation used for calculating the herd immunity threshold can be used for calculating the vaccination level needed to eliminate a disease, written as Vc.[1] Vaccines are usually imperfect however, so the effectiveness, E, of a vaccine must be accounted for:
From this equation, it can be observed that if E is less than (1 − 1/R0), then it is impossible to eliminate a disease, even if the entire population is vaccinated.[1] Similarly, waning vaccine-induced immunity, as occurs with acellular pertussis vaccines, requires higher levels of booster vaccination to sustain herd immunity.[1][21] If a disease has ceased to be endemic to a population, then natural infections no longer contribute to a reduction in the fraction of the population that is susceptible. Only vaccination contribute to this reduction.[9] The relation between vaccine coverage and effectiveness and disease incidence can be shown by subtracting the product of the effectiveness of a vaccine and the proportion of the population that is vaccinated, pv, from the herd immunity threshold equation as follows:

It can be observed from this equation that, ceteris paribus, any increase in either vaccine coverage or vaccine effectiveness, including any increase in excess of a disease's HIT, further reduces the number of cases of a disease.[9] The rate of decline in cases depends on a disease's R0, with diseases with lower R0 values experiencing sharper declines.[9] Vaccines usually possess at least one contraindication for a specific population for medical reasons, so it is vital for both effectiveness and coverage to be high so that herd immunity can be established to protect these individuals.[16][17][20] Vaccine effectiveness is often, but not always, adversely affected by passive immunity,[63][64] so additional doses are recommended for some vaccines while others are not administered until after an individual has lost his or her passive immunity.[15][20]
Passive immunity
Individual immunity can also be gained passively, in which antibodies to a pathogen are transferred from one individual to another. This can occur naturally, whereby maternal antibodies, primarily immunoglobulin G antibodies, are transferred across the placenta and in colostrum to fetuses and newborns,[65][66] or artificially, by which antibodies from the serum or plasma of an immune individual are injected into a susceptible person.[59][67] Protection generated from passive immunity is immediate but wanes over the course of weeks to months, so any contribution to herd immunity is temporary.[5][59][68] For diseases that are especially severe among fetuses and newborns, such as influenza and tetanus, pregnant women may be immunized in order to transfer antibodies to the child.[17][69][70] In the same way, high-risk groups that are either more likely to experience infection or are more likely to develop complications from infection may receive antibody preparations to prevent these infections or to reduce the severity of symptoms.[67]
Cost–benefit analysis
Herd immunity is often accounted for when conducting cost–benefit analyses of vaccination programs. It is regarded as a positive externality of high levels of immunity, producing an additional benefit of disease reduction that would not occur had no herd immunity been generated in the population.[71][72] Therefore, herd immunity's inclusion in cost–benefit analyses results in more favorable cost-effectiveness or cost–benefit ratios and an increase in the number of disease cases averted by vaccination.[72] Study designs done to estimate herd immunity's benefit include recording disease incidence in households in which a member was vaccinated, randomizing a population in a single geographic area to be vaccinated or not, and observing disease incidence before and after a vaccination program is introduced.[73] From these, it can be observed that disease incidence may decrease to a level beyond what can be predicted from direct protection alone, indicating that herd immunity contributed to the reduction.[73] When serotype replacement is accounted for, it lessens the predicted benefits of vaccination.[72]
History
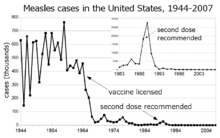
The term herd immunity was first used in 1923 to refer to an entire population's immunity, in reference to research examining disease mortality in mouse populations with varying degrees of immunity.[74] Herd immunity was first recognized as a naturally occurring phenomenon in the 1930s when A. W. Hedrich published research on the epidemiology of measles in Baltimore and took notice that after many children had become immune to measles, the number of new infections temporarily decreased, including among susceptible children.[8] In spite of this knowledge, efforts to control and eliminate measles were unsuccessful until mass vaccination using the measles vaccine began in the 1960s.[8] Mass vaccination, discussions of disease eradication, and cost–benefit analyses of vaccination subsequently prompted more widespread use of the term herd immunity.[1] In the 1970s, the theorem used to calculated a disease's herd immunity threshold was developed.[1] During the smallpox eradication campaign in the 1960s and 1970s, the practice of ring vaccination, of which herd immunity is integral to, began as a way to immunize every person in a "ring" around an infected individual to prevent outbreaks from spreading.[75]
Since the adoption of mass and ring vaccination, complexities and challenges to herd immunity have arisen.[1][56] Modelling of the spread of infectious disease originally made a number of assumptions, namely that entire populations are susceptible and well-mixed, which do not exist in reality, so more precise equations have been developed.[1] In recent decades, it has been recognized that the dominant strain of a microorganism in circulation may change due to herd immunity, either because of herd immunity acting as an evolutionary pressure or because herd immunity against one strain allowed another already-existing strain to spread.[32][38] Emerging or ongoing vaccine controversies and various reasons for opposing vaccination have made herd immunity to either not be established or to disappear in certain communities, allowing preventable diseases to persist in or return to these communities.[10][11][12]
See also
References
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Fine, P.; Eames, K.; Heymann, D. L. (1 April 2011). ""Herd immunity": A rough guide". Clinical Infectious Diseases. 52 (7): 911–6. doi:10.1093/cid/cir007. PMID 21427399.
- ↑ Gordis, L. (14 November 2013). Epidemiology. Elsevier Health Sciences. pp. 26–27. ISBN 9781455742516. Retrieved 29 March 2015.
- 1 2 3 4 Merrill, R. M. (2013). Introduction to Epidemiology. Jones & Bartlett Publishers. pp. 68–71. ISBN 9781449645175. Retrieved 29 March 2015.
- 1 2 3 4 5 "Herd Immunity". oxford vaccine group. Retrieved 24 March 2015.
- 1 2 3 4 5 6 Somerville, M.; Kumaran, K.; Anderson, R. (19 January 2012). Public Health and Epidemiology at a Glance. John Wiley & Sons. pp. 58–59. ISBN 9781118308646. Retrieved 29 March 2015.
- 1 2 3 4 Cliff, A.; Smallman-Raynor, M. (11 April 2013). Oxford Textbook of Infectious Disease Control: A Geographical Analysis from Medieval Quarantine to Global Eradication. Oxford University Press. pp. 125–136. ISBN 9780199596614. Retrieved 29 March 2015.
- 1 2 3 4 5 6 7 Kim, T. H.; Jonhstone, J.; Loeb, M. (September 2011). "Vaccine herd effect". Scandinavian Journal of Infectious Diseases. 43 (9): 683–9. doi:10.3109/00365548.2011.582247. PMC 3171704
 . PMID 21604922.
. PMID 21604922. - 1 2 3
- Hinman, A. R.; Orenstein, W. A.; Papania, M. J. (1 May 2004). "Evolution of measles elimination strategies in the United States". The Journal of Infectious Diseases. 189 (Suppl 1): S17–22. doi:10.1086/377694. PMID 15106084.
*Sencer, D. J.; Dull, H. B.; Langmuir, A. D. (March 1967). "Epidemiologic basis for eradication of measles in 1967". Public Health Reports. 82 (3): 253–6. doi:10.2307/4592985. PMC 1919891 . PMID 4960501.
. PMID 4960501.
- Hinman, A. R.; Orenstein, W. A.; Papania, M. J. (1 May 2004). "Evolution of measles elimination strategies in the United States". The Journal of Infectious Diseases. 189 (Suppl 1): S17–22. doi:10.1086/377694. PMID 15106084.
- 1 2 3 4 5 6 7 8 9 10 11 12 Garnett, G. P. (1 February 2005). "Role of Herd Immunity in Determining the Effect of Vaccines against Sexually Transmitted Disease". The Journal of Infectious Diseases. 191 (Suppl 1): S97–106. doi:10.1086/425271. PMID 15627236.
- 1 2 3 Quadri-Sheriff, M.; Hendrix, K. S.; Downs, S. M.; Sturm, L. A.; Zimet, G. D.; Finnell, S. M. (September 2012). "The role of herd immunity in parents' decision to vaccinate children: a systematic review". Pediatrics. 130 (3): 522–30. doi:10.1542/peds.2012-0140. PMID 22926181.
- 1 2 3 4 5 Dubé, E.; Laberge, C.; Guay, M.; Bramadat, P.; Roy, R.; Bettinger, J. (August 2013). "Vaccine hesitancy: an overview". Human Vaccines & Immunotherapeutics. 9 (8): 1763–73. doi:10.4161/hv.24657. PMC 3906279
 . PMID 23584253.
. PMID 23584253. - 1 2 3 4 Ropeik, D. (August 2013). "How society should respond to the risk of vaccine rejection". Human Vaccines & Immunotherapeutics. 9 (8): 1815–8. doi:10.4161/hv.25250. PMC 3906287
 . PMID 23807359.
. PMID 23807359. - ↑ Wane, Joanna. "The case for vaccination" (PDF). North & South. Bauer Media. Retrieved 3 July 2015.
- 1 2 3 4 Cesaro, S.; Giacchino, M.; Fioredda, F.; Barone, A.; Battisti, L.; Bezzio, S.; Frenos, S.; De Santis, R.; Livadiotti, S.; Marinello, S.; Zanazzo, A. G.; Caselli, D. (2014). "Guidelines on vaccinations in paediatric haematology and oncology patients". Biomed Res Int. 2014: 707691. doi:10.1155/2014/707691. PMC 4020520
 . PMID 24868544.
. PMID 24868544. - 1 2 3 National Center for Immunization and Respiratory Diseases (2011). "General recommendations on immunization --- recommendations of the Advisory Committee on Immunization Practices (ACIP)". MMWR. Recommendations and reports : Morbidity and mortality weekly report. Recommendations and reports / Centers for Disease Control. 60 (2): 1–64. PMID 21293327.
- 1 2 Wolfe, R. M. (2012). "Update on adult immunizations". The Journal of the American Board of Family Medicine. 25 (4): 496–510. doi:10.3122/jabfm.2012.04.100274. PMID 22773718.
- 1 2 3 Munoz, F. M. (2013). "Maternal immunization: An update for pediatricians". Pediatric Annals. 42 (8): 153–8. doi:10.3928/00904481-20130723-09. PMID 23910028.
- ↑ Esposito, S; Bosis, S; Morlacchi, L; Baggi, E; Sabatini, C; Principi, N (2012). "Can infants be protected by means of maternal vaccination?". Clinical Microbiology and Infection. 18 Suppl 5: 85–92. doi:10.1111/j.1469-0691.2012.03936.x. PMID 22862749.
- 1 2 3 Rakel, D.; Rakel, R. E. (2 February 2015). Textbook of Family Medicine. Elsevier Health Sciences. pp. 99, 187. ISBN 9780323313087. Retrieved 30 March 2015.
- 1 2 3 Tulchinsky, T. H.; Varavikova, E. A. (26 March 2014). The New Public Health: An Introduction for the 21st Century. Academic Press. pp. 163–182. ISBN 9780124157675. Retrieved 30 March 2015.
- 1 2 McGirr, A; Fisman, D. N. (2015). "Duration of Pertussis Immunity After DTaP Immunization: A Meta-analysis". Pediatrics. 135 (2): 331–343. doi:10.1542/peds.2014-1729. PMID 25560446.
- ↑ Zepp, F; Heininger, U; Mertsola, J; Bernatowska, E; Guiso, N; Roord, J; Tozzi, A. E.; Van Damme, P (2011). "Rationale for pertussis booster vaccination throughout life in Europe". The Lancet Infectious Diseases. 11 (7): 557–70. doi:10.1016/S1473-3099(11)70007-X. PMID 21600850.
- 1 2 3 4 5 6 Pittet, L. F.; Posfay-Barbe, K. M. (2012). "Pneumococcal vaccines for children: A global public health priority". Clinical Microbiology and Infection. 18 Suppl 5: 25–36. doi:10.1111/j.1469-0691.2012.03938.x. PMID 22862432.
- ↑ Nakagomi, O; Iturriza-Gomara, M; Nakagomi, T; Cunliffe, N. A. (2013). "Incorporation of a rotavirus vaccine into the national immunisation schedule in the United Kingdom: A review". Expert Opinion on Biological Therapy. 13 (11): 1613–21. doi:10.1517/14712598.2013.840285. PMID 24088009.
- ↑ Lopman, B. A.; Payne, D. C.; Tate, J. E.; Patel, M. M.; Cortese, M. M.; Parashar, U. D. (2012). "Post-licensure experience with rotavirus vaccination in high and middle income countries; 2006 to 2011". Current Opinion in Virology. 2 (4): 434–42. doi:10.1016/j.coviro.2012.05.002. PMID 22749491.
- 1 2 Kim, T. H. (2014). "Seasonal influenza and vaccine herd effect". Clinical and Experimental Vaccine Research. 3 (2): 128–32. doi:10.7774/cevr.2014.3.2.128. PMC 4083064
 . PMID 25003085.
. PMID 25003085. - 1 2 3 Lowy, D. R.; Schiller, J. T. (2012). "Reducing HPV-associated cancer globally". Cancer Prevention Research. 5 (1): 18–23. doi:10.1158/1940-6207.CAPR-11-0542. PMC 3285475
 . PMID 22219162.
. PMID 22219162. - 1 2 3 4 5 Lenzi, A; Mirone, V; Gentile, V; Bartoletti, R; Ficarra, V; Foresta, C; Mariani, L; Mazzoli, S; Parisi, S. G.; Perino, A; Picardo, M; Zotti, C. M. (2013). "Rome Consensus Conference - statement; human papilloma virus diseases in males". BMC Public Health. 13: 117. doi:10.1186/1471-2458-13-117. PMC 3642007
 . PMID 23391351.
. PMID 23391351. - ↑ Garland, S. M.; Skinner, S. R.; Brotherton, J. M. (2011). "Adolescent and young adult HPV vaccination in Australia: Achievements and challenges". Preventive Medicine. 53 Suppl 1: S29–35. doi:10.1016/j.ypmed.2011.08.015. PMID 21962468.
- 1 2 3 4 5 6 7 8 Rodpothong, P; Auewarakul, P (2012). "Viral evolution and transmission effectiveness". World Journal of Virology. 1 (5): 131–4. doi:10.5501/wjv.v1.i5.131. PMC 3782273
 . PMID 24175217.
. PMID 24175217. - 1 2 Corti, D; Lanzavecchia, A (2013). "Broadly neutralizing antiviral antibodies". Annual Review of Immunology. 31: 705–42. doi:10.1146/annurev-immunol-032712-095916. PMID 23330954.
- 1 2 3 Bull, R. A.; White, P. A. (2011). "Mechanisms of GII.4 norovirus evolution". Trends in Microbiology. 19 (5): 233–40. doi:10.1016/j.tim.2011.01.002. PMID 21310617.
- 1 2 Ramani, S; Atmar, R. L.; Estes, M. K. (2014). "Epidemiology of human noroviruses and updates on vaccine development". Current Opinion in Gastroenterology. 30 (1): 25–33. doi:10.1097/MOG.0000000000000022. PMC 3955997
 . PMID 24232370.
. PMID 24232370. - 1 2 3 Pleschka, S (2013). "Overview of Influenza Viruses". Swine Influenza. Current Topics in Microbiology and Immunology. 370. pp. 1–20. doi:10.1007/82_2012_272. ISBN 978-3-642-36870-7. PMID 23124938.
- ↑ Han, T; Marasco, W. A. (2011). "Structural basis of influenza virus neutralization". Annals of the New York Academy of Sciences. 1217: 178–90. doi:10.1111/j.1749-6632.2010.05829.x. PMC 3062959
 . PMID 21251008.
. PMID 21251008. - ↑ Reperant, L. A.; Rimmelzwaan, G. F.; Osterhaus, A. D. (2014). "Advances in influenza vaccination". F1000prime reports. 6: 47. doi:10.12703/p6-47. PMC 4047948
 . PMID 24991424.
. PMID 24991424. - 1 2 3 Weinberger, D. M.; Malley, R; Lipsitch, M (2011). "Serotype replacement in disease after pneumococcal vaccination". The Lancet. 378 (9807): 1962–73. doi:10.1016/S0140-6736(10)62225-8. PMC 3256741
 . PMID 21492929.
. PMID 21492929. - 1 2 3 McEllistrem, M. C.; Nahm, M. H. (2012). "Novel pneumococcal serotypes 6C and 6D: Anomaly or harbinger". Clinical Infectious Diseases. 55 (10): 1379–86. doi:10.1093/cid/cis691. PMC 3478140
 . PMID 22903767.
. PMID 22903767. - ↑ Dagan, R (2009). "Impact of pneumococcal conjugate vaccine on infections caused by antibiotic-resistant Streptococcus pneumoniae". Clinical Microbiology and Infection. 15 Suppl 3: 16–20. doi:10.1111/j.1469-0691.2009.02726.x. PMID 19366365.
- ↑ Lynch Jp, 3rd; Zhanel, G. G. (2010). "Streptococcus pneumoniae: Epidemiology and risk factors, evolution of antimicrobial resistance, and impact of vaccines". Current Opinion in Pulmonary Medicine. 16 (3): 217–25. doi:10.1097/MCP.0b013e3283385653. PMID 20375783.
- ↑ Njeumi, F; Taylor, W; Diallo, A; Miyagishima, K; Pastoret, P. P.; Vallat, B; Traore, M (2012). "The long journey: A brief review of the eradication of rinderpest". Revue scientifique et technique (International Office of Epizootics). 31 (3): 729–46. PMID 23520729.
- 1 2 Smith, K. A. (2013). "Smallpox: Can we still learn from the journey to eradication?". The Indian journal of medical research. 137 (5): 895–9. PMC 3734679
 . PMID 23760373.
. PMID 23760373. - 1 2 3 4 5 Perisic, A; Bauch, C. T. (2009). "Social contact networks and disease eradicability under voluntary vaccination". PLoS Computational Biology. 5 (2): e1000280. doi:10.1371/journal.pcbi.1000280. PMC 2625434
 . PMID 19197342.
. PMID 19197342. - 1 2 3 4 5 6 7 Fu, F; Rosenbloom, D. I.; Wang, L; Nowak, M. A. (2011). "Imitation dynamics of vaccination behaviour on social networks". Proceedings of the Royal Society B: Biological Sciences. 278 (1702): 42–9. doi:10.1098/rspb.2010.1107. PMC 2992723
 . PMID 20667876.
. PMID 20667876. - ↑ Wicker, S; Maltezou, H. C. (2014). "Vaccine-preventable diseases in Europe: Where do we stand?". Expert Review of Vaccines. 13 (8): 979–87. doi:10.1586/14760584.2014.933077. PMID 24958075.
- 1 2 3 Fukuda, E.; Tanimoto, J. (4 November 2014). Impact of Stubborn Individuals on a Spread of Infectious Disease under Voluntary Vaccination Policy. Springer. pp. 1–10. ISBN 9783319133591. Retrieved 30 March 2015.
- 1 2 Barrett, Scott (15 December 2014). "Global Public Goods and International Development". In J. Warren Evans, Robin Davies. Too Global To Fail: The World Bank at the Intersection of National and Global Public Policy in 2025. World Bank Publications. pp. 13–18. ISBN 978-1-4648-0310-9.
- ↑ Parker, A. M.; Vardavas, R; Marcum, C. S.; Gidengil, C. A. (2013). "Conscious consideration of herd immunity in influenza vaccination decisions". American Journal of Preventive Medicine. 45 (1): 118–21. doi:10.1016/j.amepre.2013.02.016. PMC 3694502
 . PMID 23790997.
. PMID 23790997. - 1 2 3 Gowda, C; Dempsey, A. F. (2013). "The rise (and fall?) of parental vaccine hesitancy". Human Vaccines & Immunotherapeutics. 9 (8): 1755–62. doi:10.4161/hv.25085. PMC 3906278
 . PMID 23744504.
. PMID 23744504. - ↑ Ozawa, S; Stack, M. L. (2013). "Public trust and vaccine acceptance--international perspectives". Human Vaccines & Immunotherapeutics. 9 (8): 1774–8. doi:10.4161/hv.24961. PMC 3906280
 . PMID 23733039.
. PMID 23733039. - ↑ Unless noted, R0 values are from: History and Epidemiology of Global Smallpox Eradication From the training course titled "Smallpox: Disease, Prevention, and Intervention". The Centers for Disease Control and Prevention and the World Health Organization. Slide 17. Retrieved 13 March 2015.
- 1 2 Biggerstaff, M; Cauchemez, S; Reed, C; Gambhir, M; Finelli, L (2014). "Estimates of the reproduction number for seasonal, pandemic, and zoonotic influenza: A systematic review of the literature". BMC Infectious Diseases. 14: 480. doi:10.1186/1471-2334-14-480. PMC 4169819
 . PMID 25186370.
. PMID 25186370. - ↑ Wallinga, J; Teunis, P (2004). "Different epidemic curves for severe acute respiratory syndrome reveal similar impacts of control measures". American Journal of Epidemiology. 160 (6): 509–16. doi:10.1093/aje/kwh255. PMID 15353409.
- ↑ Althaus, C. L. (2014). "Estimating the Reproduction Number of Ebola Virus (EBOV) During the 2014 Outbreak in West Africa". PLoS Currents. 6. doi:10.1371/currents.outbreaks.91afb5e0f279e7f29e7056095255b288. PMC 4169395
 . PMID 25642364.
. PMID 25642364. - 1 2 Dabbaghian, V.; Mago, V. K. (27 October 2013). Theories and Simulations of Complex Social Systems. Springer. pp. 134–135. ISBN 978-3-642-39149-1. Retrieved 29 March 2015.
- 1 2 3 Rashid, H; Khandaker, G; Booy, R (2012). "Vaccination and herd immunity: What more do we know?". Current Opinion in Infectious Diseases. 25 (3): 243–9. doi:10.1097/QCO.0b013e328352f727. PMID 22561998.
- ↑ Maglione, M. A.; Das, L; Raaen, L; Smith, A; Chari, R; Newberry, S; Shanman, R; Perry, T; Goetz, M. B.; Gidengil, C (2014). "Safety of vaccines used for routine immunization of U.S. Children: A systematic review". Pediatrics. 134 (2): 325–37. doi:10.1542/peds.2014-1079. PMID 25086160.
- ↑ Demicheli, V; Rivetti, A; Debalini, M. G.; Di Pietrantonj, C (2012). "Vaccines for measles, mumps and rubella in children". Cochrane Database of Systematic Reviews. 2. pp. CD004407. doi:10.1002/14651858.CD004407.pub3. PMID 22336803.
- 1 2 3 Pommerville, J. C. (2 December 2014). Fundamentals of Microbiology: Body Systems Edition. Jones & Bartlett Publishers. pp. 559–563. ISBN 9781284057102. Retrieved 30 March 2015.
- ↑ Papaloukas, O; Giannouli, G; Papaevangelou, V (2014). "Successes and challenges in varicella vaccine". Therapeutic Advances in Vaccines. 2 (2): 39–55. doi:10.1177/2051013613515621. PMC 3991154
 . PMID 24757524.
. PMID 24757524. - ↑ Shann, F (2013). "Nonspecific effects of vaccines and the reduction of mortality in children". Clinical Therapeutics. 35 (2): 109–14. doi:10.1016/j.clinthera.2013.01.007. PMID 23375475.
- ↑ Visser, A; Hoosen, A (2012). "Haemophilus influenzae type b conjugate vaccines - a South African perspective". Vaccine. 30 Suppl 3: C52–7. doi:10.1016/j.vaccine.2012.06.022. PMID 22939022.
- ↑ Leuridan, E; Sabbe, M; Van Damme, P (2012). "Measles outbreak in Europe: Susceptibility of infants too young to be immunized". Vaccine. 30 (41): 5905–13. doi:10.1016/j.vaccine.2012.07.035. PMID 22841972.
- ↑ Hodgins, D. C.; Shewen, P. E. (2012). "Vaccination of neonates: Problem and issues". Vaccine. 30 (9): 1541–59. doi:10.1016/j.vaccine.2011.12.047. PMID 22189699.
- ↑ Chucri, T. M.; Monteiro, J. M.; Lima, A. R.; Salvadori, M. L.; Kfoury Jr, J. R.; Miglino, M. A. (2010). "A review of immune transfer by the placenta". Journal of Reproductive Immunology. 87 (1-2): 14–20. doi:10.1016/j.jri.2010.08.062. PMID 20956021.
- ↑ Palmeira, P; Quinello, C; Silveira-Lessa, A. L.; Zago, C. A.; Carneiro-Sampaio, M (2012). "IgG placental transfer in healthy and pathological pregnancies". Clinical and Developmental Immunology. 2012: 985646. doi:10.1155/2012/985646. PMC 3251916
 . PMID 22235228.
. PMID 22235228. - 1 2 Parija, S. C. (10 February 2014). Textbook of Microbiology & Immunology. Elsevier Health Sciences. pp. 88–89. ISBN 9788131236246. Retrieved 30 March 2015.
- ↑ Detels, R.; Gulliford, M.; Karim, Q. A.; Tan, C. C. (1 February 2015). Oxford Textbook of Global Public Health. Oxford University Press. p. 1490. ISBN 9780199661756. Retrieved 30 March 2015.
- ↑ Demicheli, V; Barale, A; Rivetti, A (2013). "Vaccines for women to prevent neonatal tetanus". Cochrane Database of Systematic Reviews. 5. pp. CD002959. doi:10.1002/14651858.CD002959.pub3. PMID 23728640.
- ↑ Swamy, G. K.; Garcia-Putnam, R (2013). "Vaccine-preventable diseases in pregnancy". American Journal of Perinatology. 30 (2): 89–97. doi:10.1055/s-0032-1331032. PMID 23271378.
- ↑ Bärnighausen, T.; Bloom, D. E.; Cafiero-Fonseca, E. T.; O'Brien, J. C. (26 August 2014). "Valuing vaccination". Proc Natl Acad Sci U S A. 111 (34): 12313–9. doi:10.1073/pnas.1400475111. PMC 4151736
 . PMID 25136129.
. PMID 25136129. - 1 2 3 Deogaonkar, R.; Hutubessy, R.; van der Putten I.; Evers S.; Jit M. (16 October 2012). "Systematic review of studies evaluating the broader economic impact of vaccination in low and middle income countries". BMC Public Health. 12: 878. doi:10.1186/1471-2458-12-878. PMC 3532196
 . PMID 23072714.
. PMID 23072714. - 1 2 Jit, M.; Newall, A. T.; Beutels, P. (April 2013). "Key issues for estimating the impact and cost-effectiveness of seasonal influenza vaccination strategies". Hum Vaccin Immunother. 9 (4): 834–40. doi:10.4161/hv.23637. PMC 3903903
 . PMID 23357859.
. PMID 23357859. - ↑ Topley, W. W. C.; Wilson, G. S. (May 1923). "The Spread of Bacterial Infection. The Problem of Herd-Immunity". The Journal of Hygiene (London). 21 (3): 243–249. PMC 2167341
 .
. - ↑ Strassburg, M. A. (1982). "The global eradication of smallpox". American journal of infection control. 10 (2): 53–9. doi:10.1016/0196-6553(82)90003-7. PMID 7044193.
External links
- A visual simulation of herd immunity written by Shane Killian and modified by Robert Webb