Frequency (gene)
| Frequency clock protein | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | FRQ | ||||||||
| Pfam | PF09421 | ||||||||
| InterPro | IPR018554 | ||||||||
| |||||||||
Frequency (frq) is a gene that encodes the frequency protein (FRQ) found in the Neurospora crassa circadian clock. FRQ plays a key role in the auto regulatory transcription-translation negative feedback-loop (TTFL), which generates daily rhythms in gene expression.[1]
Discovery
Colin Pittendrigh and his colleagues discovered the presence of a circadian clock in Neurospora crassa in 1959.[2] Malcolm L. Sargent, Winslow R. Briggs and Dow O. Woodward at Stanford University later discovered circadian rhythms in conidiation in a strain of Neurospora called Timex.[3] This strain contained a mutation in the locus band (bd) which was responsible for the circadian spore formation.[4] Strains including the bd locus are now used for all studies of circadian biology in Neurospora.[4] Mutant genes frq-1, frq-2, and frq-3 were discovered by Jerry F. Feldman and Marian N. Hoyle in 1973.[5] In 1986, frq was first cloned by Jay Dunlap and his colleagues; its expression was later shown to rhythmically cycle.[6] This sparked interest in further research and understanding of the N. crassa circadian clock.[7]
Function
Frq’s important circadian function is supported by experiments which show that deletion of the frq gene results in arrhythmicity. Frq forms two transcripts that encode two different FRQ proteins: a long form of 989 amino acids (lFRQ) and a short form of 890 amino acids (sFRQ).[9] An increase in temperature leads to increased expression of lFRQ, while sFRQ is unaffected. Warmer temperatures induce more efficient splicing of an intron in the translation start site.[10] Both lFRQ and sFRQ are required for strong rhythmicity although the clock is able to persist at certain temperatures, albeit with a weaker rhythmicity, with just one of the proteins present.[11] Since sFRQ favors a longer period than lFRQ, free running rhythms in wild type Neurospora are somewhat decreased with increased temperature.[10] FRQ protein has also been shown to interact with FRH (FRQ-interacting RNA helicase; an essential DEAD box-containing RNA helicase in Neurospora) to form a FRQ/FRH complex (FFC).[12] Overexpression of FRQ protein has been shown to inhibit frq expression, which eventually led to the discovery of the presence of a transcription translation negative feedback loop system in Neurospora.[13] Outputs of the Neurospora circadian clock include carotenoid synthesis and spore formation.[10]
Regulation
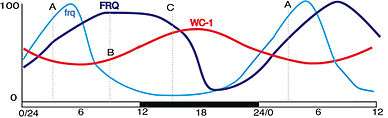
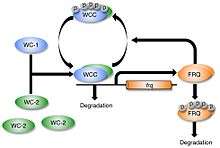
White collar-1 (WC-1) and white collar-2 (WC-2) are GATA transcription factors in Neurospora. Together, they form a heterodimeric "white collar complex" (WCC) via their PAS domains.[15] When WCC is hypophosphorylated during subjective night, it binds to the frequency (frq) gene promoter and activates frq transcription.[16] The frequency (FRQ) protein accumulates and is progressively phosphorylated by casein kinase 1 (CKI), casein kinase 2 (CKII), and a calcium/calmodulin-dependent kinase (CAMK-1), reaching its peak around mid-subjective day.[17][18] Kinase inhibitors reduce degradation of FRQ by preventing phosphorylation.[19]
FRQ forms a homodimer to interact with the white collar complex and repress its own expression.[20] Deletion of the WCC leads to an inability to form the homodimer, which causes frq to no longer be negatively regulated by FRQ concentration.[21] This leads to arrythmicity.[21] On the other hand, when FRQ is phosphorylated, it is ubiquitinated and then degraded by the FWD-1 protein, which is part of the SCF type E3 ligase.[10][22] FRQ recruits kinases such as casein kinase 1a (CK-1a) that phosphorylate WCC. Hyperphosphorylated WCC is inactive and binds poorly to the frq promoter, which inhibits frq expression. Eventually repression is relieved to allow frq transcription to continue and frq mRNA to build up again. This process occurs with a periodicity of around 22 hours in constant conditions.[23]
The frq gene is strongly induced by short duration exposure to light. Since the core of the clock is based on rhythmic expression of frq, acute light-induction provides a straightforward way to reset the clock. Mammalian clocks are reset by light by a nearly identical mechanism, with mPer1 transcripts being induced by short flashes of light outside of the subjective day. The mPer1 mechanism in the mammalian clock draws closer similarities to the mechanism in Neurospora than to the mechanism of its homolog in Drosophila, per.[24]
Mutations
Forward genetics has been used to create Neurospora clock mutants with varied periods of conidiation.[25] Mutations have been identified that cause both long and short periods relative to the wild-type value of 21.5 hours. Mutants attain a shorter or longer period due to the rate at which they are phosphorylated. Mutants that can reduce phosphorylation, due to a mutation in a binding site, do not degrade as rapidly and thus have a longer period. The reverse is true for mutants with a short period.[26] The periods of various frq mutants at 25oC are as follows:

| Mutant | frq-1 | frq-2 | frq-3 | frq-4 | frq-6 | frq-7 | frq-8 | frq-9 |
| Period (hr) | 16.5 | 19.3 | 24.0 | 19.3 | 19.2 | 29.0 | 29.0 | Arrhythmic |
Frq-1, frq-2, and frq-3 are very closely linked since no genetic recombination among them is detected. Each mutant segregates as a single nuclear gene.[27] Of these mutants, the mutations that lead to a shorter period (frq-1, frq-2, frq-3, frq-6) do not affect the temperature compensation response. However, the mutants that result in a longer period (frq-3, frq-7, frq-8) show signs of a lower breakpoint temperature, with the lowest breakpoint temperature corresponding to the longest period (29.0 hr).[28] In addition, one recessive allele, frq-9 results in conditional arrhythmicity and a complete loss of temperature compensation.[29]
FRQ-less Oscillator (FLO)
Oscillators outside of the FRQ/WCC system have been discovered; however, these FRQ-less oscillations do not satisfy the characteristics to be classified as circadian oscillators.[30] The FWO has been shown, via luciferase reporting, to continue running even when the FLO controls conidiation.[30] This has been shown in strains lacking choline.[30] In frq-9 mutant Neurospora crassa, a non-temperature compensated rhythm of conidiospore development was still observed in constant darkness (DD).[31] The period for frq null mutants varied from 12 to 35 hours but could be stabilized by the addition of farnesol or geraniol. However, this mechanism is not well understood.[32] Although the FRQ-less rhythm lost certain clock characteristics such as temperature compensation, temperature pulses were sufficient to reset the clock.[33] There is evidence to support FRQ-less oscillators in Neurospora crassa. For example, many are "slaves" to the frequency/white collar oscillator since they do not possess all of the characteristics of a circadian clock on their own.[32] However, rhythms in clock-controlled gene-16 (ccg-16) are coupled to the FWO but function autonomously, demonstrating that Neurospora crassa contains at least 2 potential pacemakers, but only one that can be reset by light temperature while maintaining temperature compensation.[32][34] The FRQ-less oscillator has never been proven to affect the true circadian clock.[34] The mechanism and significance for FRQ-less oscillators (FLO) are still under research.
Evolution
Since codon optimization of the frq gene results in impaired circadian feedback loop function, frq displays non-optimal codon usage bias across its open reading frame in contrast to most other genes.[35] FRQ is an intrinsically disordered protein that is not well conserved, even across fungi.[36][37] However, WC1 is very well conserved as a blue light photoreceptor, similar in structure and function to BMAL1.[36] Casein kinase 2 is conserved in the circadian oscillators of plants (Arabidopsis) and flies (Drosophila).[18] A similar form of casein kinase 1 is necessary for the degradation of period (PER) proteins in Drosophila and mammals.[18] The Drosophila gene slimb is orthologous to FWD1 in Neurospora, both of which are crucial for clock protein degradation.[18]
References
- ↑ Baker, Christopher L.; Loros, Jennifer J.; Dunlap, Jay C. (2012-01-01). "The circadian clock of Neurospora crassa". FEMS Microbiology Reviews. 36 (1): 95–110. doi:10.1111/j.1574-6976.2011.00288.x. ISSN 1574-6976. PMID 21707668. Retrieved 2015-04-22.
- ↑ Pittendrigh, C. S.; Bruce, V. G.; Rosensweig, N. S.; Rubin, M. L. (July 18, 1959). "Growth Patterns in Neurospora: A Biological Clock in Neurospora". Nature. 184 (4681): 169–170. doi:10.1038/184169a0. Retrieved 2015-04-22.
- ↑ Sargent ML, Briggs WR, Woodward DO (October 1966). "Circadian nature of a rhythm expressed by an invertaseless strain of Neurospora crassa". Plant Physiol. 41 (8): 1343–9. doi:10.1104/pp.41.8.1343. PMC 550529
 . PMID 5978549.
. PMID 5978549. - 1 2 Belden, W. J., L. F. Larrondo, A. C. Froehlich, M. Shi, C.-H. Chen, J. J. Loros, and J. C. Dunlap. "The Band Mutation in Neurospora Crassa Is a Dominant Allele of Ras-1 Implicating RAS Signaling in Circadian Output." Genes & Development 21.12 (2007): 1494-505. Web.
- ↑ Feldman JF, Hoyle MN (December 1973). "Isolation of circadian clock mutants of Neurospora crassa". Genetics. 75 (4): 605–13. PMC 1213033
 . PMID 4273217.
. PMID 4273217. - ↑ Aronson, B. D.; Johnson, K. A.; Loros, J. J.; Dunlap, J. C. (Mar 18, 1994). "Negative feedback defining a circadian clock: autoregulation of the clock gene frequency". Science. 263 (5153): 1578–1584. doi:10.1126/science.8128244. ISSN 0036-8075. PMID 8128244.
- ↑ McClung CR, Fox BA, Dunlap JC (June 1989). "The Neurospora clock gene Frequency shares a sequence element with the Drosophila clock gene period". Nature. 339 (6225): 558–62. doi:10.1038/339558a0. PMID 2525233.
- ↑ Tseng YY, Hunt SM, Heintzen C, Crosthwaite SK, Schwartz JM (2012). "Comprehensive modelling of the Neurospora circadian clock and its temperature compensation". PLoS Comput. Biol. 8 (3): e1002437. doi:10.1371/journal.pcbi.1002437. PMC 3320131
 . PMID 22496627.
. PMID 22496627. - ↑ Nakashima H, Onai K (December 1996). "The circadian conidiation rhythm in Neurospora crassa". Seminars in Cell & Developmental Biology. 7 (6): 765–774. doi:10.1006/scdb.1996.0094.
- 1 2 3 4 Diernfellner, Axel; Colot, Hildur V.; Dintsis, Orfeas; Loros, Jennifer J.; Dunlap, Jay C.; Brunner, Michael. "Long and short isoforms of Neurospora clock protein FRQ support temperature-compensated circadian rhythms". FEBS Letters. 581 (30): 5759–5764. doi:10.1016/j.febslet.2007.11.043.
- ↑ Liu Y, Garceau NY, Loros JJ, Dunlap JC (May 1997). "Thermally regulated translational control of FRQ mediates aspects of temperature responses in the neurospora circadian clock". Cell. 89 (3): 477–86. doi:10.1016/S0092-8674(00)80228-7. PMID 9150147.
- ↑ Cheng P, He Q, He Q, Wang L, Liu Y (January 2005). "Regulation of the Neurospora circadian clock by an RNA helicase". Genes Dev. 19 (2): 234–41. doi:10.1101/gad.1266805. PMC 545885
 . PMID 15625191.
. PMID 15625191. - ↑ Young, MW; Kay, SA (September 2001). "Time zones: a comparative genetics of circadian clocks". Nature Reviews Genetics. 2: 702–715. doi:10.1038/35088576. PMID 11533719.
- ↑ "A Circadian Clock in Neurospora: How Genes and Proteins Cooperate to Produce a Sustained, Entrainable, and Compensated Biological Oscillator with a Period of about a Day". Cold Spring Harbor Symposia on Quantitative Biology. 72: 57–68. doi:10.1101/sqb.2007.72.072.
- ↑ Talora, C.; et al. (1999). "Role of a white collar‐1–white collar‐2 complex in blue‐light signal transduction". The EMBO Journal. 18 (18): 4961–4968. doi:10.1093/emboj/18.18.4961. PMC 1171567
 . PMID 10487748.
. PMID 10487748. - ↑ Denault, Deanna L.; Loros, Jennifer J.; Dunlap, Jay C. (2001). "WC‐2 mediates WC‐1–FRQ interaction within the PAS protein‐linked circadian feedback loop of Neurospora". The EMBO Journal. 20 (1-2): 109–117. doi:10.1093/emboj/20.1.109.
- ↑ Heintzen, Christian; Liu, Yi (2007). "The Neurospora crassa circadian clock". Advances in Genetics. 58: 25–66. doi:10.1016/s0065-2660(06)58002-2.
- 1 2 3 4 He, Q.; Cheng, P.; Yang, Y.; He, Q.; Yu, H.; Liu, Y. (2003). "FWD1-mediated degradation of FREQUENCY in Neurospora establishes a conserved mechanism for circadian clock regulation". The EMBO Journal. 22: 4421–4430. doi:10.1093/emboj/cdg425.
- ↑ Liu, Yi; Loros, Jennifer; Dunlap, Jay C. (2000). "Phosphorylation of the Neurospora clock protein FREQUENCY determines its degradation rate and strongly influences the period length of the circadian clock". Proceedings of the National Academy of Sciences. 97 (1): 234–239. doi:10.1073/pnas.97.1.234. PMC 26646
 . PMID 10618401.
. PMID 10618401. - ↑ Cheng, Ping; et al. (2001). "Coiled‐coil domain‐mediated FRQ–FRQ interaction is essential for its circadian clock function in Neurospora". The EMBO Journal. 20 (1-2): 101–108. doi:10.1093/emboj/20.1.101. PMC 140186
 . PMID 11226160.
. PMID 11226160. - 1 2 Cheng, Ping; Yang, Yuhong; Heintzen, Christian; Liu, Yi (2001-1-15). "Coiled-coil domain-mediated FRQ–FRQ interaction is essential for its circadian clock function in Neurospora". The EMBO Journal. 20 (1-2): 101–108. doi:10.1093/emboj/20.1.101. ISSN 0261-4189. PMC 140186
 . PMID 11226160. Check date values in:
. PMID 11226160. Check date values in: |date=(help) - ↑ Salichos, L; Rokas, A (2010). "The diversity and evolution of circadian clock proteins in Fungi". Mycologia. 102: 269–78. doi:10.3852/09-073.
- ↑ Larrondo, Luis F.; et al. (2015). "Decoupling circadian clock protein turnover from circadian period determination". Science. 347 (6221): 1257277. doi:10.1126/science.1257277.
- ↑ Shigeyoshi, Yasufumi; et al. (1997). "Light-Induced Resetting of a Mammalian Circadian Clock Is Associated with Rapid Induction of the mPer1Transcript". Cell. 91 (7): 1043–1053. doi:10.1016/s0092-8674(00)80494-8. PMID 9428526.
- ↑ Merrow, Martha; Dragovic, Zdravko; Tan, Ying; Meyer, Gundela; Sveric, Kruno; Mason, Moyra; Ricken, Jan; Roenneberg, Till (January 1, 2003). "Combining Theoretical and Experimental Approaches to Understand the Circadian Clock". Chronobiology International. 20 (4): 559–575. doi:10.1081/CBI-120023678. ISSN 0742-0528. Retrieved 2015-04-21.
- ↑ Liu Y, Loros J, Dunlap JC (January 2000). "Phosphorylation of the Neurospora clock protein FREQUENCY determines its degradation rate and strongly influences the period length of the circadian clock". Proc. Natl. Acad. Sci. U.S.A. 97 (1): 234–9. doi:10.1073/pnas.97.1.234. PMC 26646
 . PMID 10618401.
. PMID 10618401. - ↑ Dunlap JC (1996). "Genetics and molecular analysis of circadian rhythms". Annu. Rev. Genet. 30: 579–601. doi:10.1146/annurev.genet.30.1.579. PMID 8982466.
- ↑ Gardner, George F.; Feldman, Jerry F. (June 1981). "Temperature Compensation of Circadian Period Length in Clock Mutants of Neurospora crassa" (PDF). Plant Physiology. American Society of Plant Biologists. 68: 1244–1248. doi:10.1104/pp.68.6.1244. PMID 16662086.
- ↑ Lewis MT, Feldman JF (November 1996). "Evolution of the Frequency (frq) clock locus in Ascomycete fungi" (PDF). Mol. Biol. Evol. 13 (9): 1233–41. doi:10.1093/oxfordjournals.molbev.a025689. PMID 8896376.
- 1 2 3 Shi, Mi; Larrondo, Luis F.; Loros, Jennifer J.; Dunlap, Jay C. (12/11/2007). "A developmental cycle masks output from the circadian oscillator under conditions of choline deficiency in Neurospora". Proceedings of the National Academy of Sciences. 104 (50): 20102–20107. doi:10.1073/pnas.0706631104. ISSN 0027-8424. PMID 18056807. Retrieved 2015-04-21. Check date values in:
|date=(help) - ↑ Loros, Jennifer J.; Richman, Adam; Feldman, Jerry F. (1986-12). "A Recessive Circadian Clock Mutation at the frq Locus of NEUROSPORA CRASSA". Genetics. 114 (4): 1095–1110. ISSN 0016-6731. PMC 1203030
 . PMID 2948874. Check date values in:
. PMID 2948874. Check date values in: |date=(help) - 1 2 3 Bell-Pedersen, Deborah; Cassone, Vincent M.; Earnest, David J.; Golden, Susan S.; Hardin, Paul E.; Thomas, Terry L.; Zoran, Mark J. (2005). "Circadian Rhythms from Multiple Oscillators: Lessons from Diverse Organisms". Nature Reviews Genetics. 6 (7): 544–56. doi:10.1038/nrg1633. PMC 2735866
 . PMID 15951747.
. PMID 15951747. - ↑ Granshaw T, Tsukamoto M, Brody S (August 2003). "Circadian rhythms in Neurospora crassa: farnesol or geraniol allow expression of rhythmicity in the otherwise arrhythmic strains frq10, wc-1, and wc-2". J. Biol. Rhythms. 18 (4): 287–96. doi:10.1177/0748730403255934. PMID 12932081.
- 1 2 Dunlap, Jay C.; Loros, Jennifer J. (Oct 2004). "The neurospora circadian system". Journal of Biological Rhythms. 19 (5): 414–424. doi:10.1177/0748730404269116. ISSN 0748-7304. PMID 15534321.
- ↑ Zhou M, Guo J, Cha J, Chae M, Chen S, Barral JM, Sachs MS, Liu Y (March 2013). "Non-optimal codon usage affects expression, structure and function of clock protein FRQ". Nature. 495 (7439): 111–5. doi:10.1038/nature11833. PMID 23417067.
- 1 2 "How fungi keep time: circadian system in Neurospora and other fungi". linkinghub.elsevier.com. 9: 579–587. Dec 2006. doi:10.1016/j.mib.2006.10.008. PMID 17064954. Retrieved 2015-04-21.
- ↑ "Conserved RNA Helicase FRH Acts Nonenzymatically to Support the Intrinsically Disordered Neurospora Clock Protein FRQ". linkinghub.elsevier.com. 52: 832–843. doi:10.1016/j.molcel.2013.11.005. Retrieved 2015-04-21.