Dichlorofluoromethane
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Dichloro(fluoro)methane | |
| Other names
Dichlorofluoromethane Fluorodichloromethane Monofluorodichloromethane Dichloromonofluoromethane Freon 21 Refrigerant 21 R 21 HCFC 21 Algofrene Type 5 Arcton 7 Halon 112 UN 1029 Genetron 21 | |
| Identifiers | |
| 75-43-4 | |
| 3D model (Jmol) | Interactive image |
| ChEMBL | ChEMBL116813 |
| ChemSpider | 6130 |
| ECHA InfoCard | 100.000.791 |
| EC Number | 200-869-8 |
| PubChem | 6370 |
| RTECS number | PA8400000 |
| UNII | 7GAO4CRJ0B |
| |
| |
| Properties | |
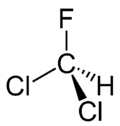
| CHCl2F | |
| Molar mass | 102.92 g/mol |
| Appearance | Colorless gas |
| Odor | ether-like[1] |
| Density | 1.405 g/cm3 at 9 °C
1.366 kg/m3 at 25 °C |
| Melting point | −135 °C (−211 °F; 138 K) |
| Boiling point | 8.92 °C (48.06 °F; 282.07 K) |
| 9.420 g/l at 30 °C | |
| log P | 1.55 |
| Vapor pressure | 160 kPa |
| Henry's law constant (kH) |
0.19 mol.kg−1.bar−1 |
| Hazards | |
| Main hazards | Dangerous for the environment (N) |
| R-phrases | R59 |
| S-phrases | S59 |
| Flash point | nonflammable [1] |
| 522 °C (972 °F; 795 K) | |
| Lethal dose or concentration (LD, LC): | |
| LC50 (median concentration) |
>800,000 mg/m3 (mouse, 2 hr) 49,900 ppm (rat, 4 hr)[2] |
| LCLo (lowest published) |
100,000 ppm (guinea pig, <1 hr) 100,000 ppm (mouse, <1 hr)[2] |
| US health exposure limits (NIOSH): | |
| PEL (Permissible) |
TWA 1000 ppm (4200 mg/m3)[1] |
| REL (Recommended) |
TWA 10 ppm (40 mg/m3)[1] |
| IDLH (Immediate danger) |
5000 ppm[1] |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Dichlorofluoromethane or Freon 21 or R 21 is a halomethane or hydrochlorofluorocarbon. It is a colorless and odorless gas.
Its critical point is at 178.5 °C (451.7 K) and 5.17 MPa (51.7 bar). At temperatures from 5 K to 105 K it has one phase in the space group Pbca.
Uses
Dichlorofluoromethane was used as a propellant and refrigerant, but due to its ozone depletion it has been set to be phased out. It has ozone depletion potential 0.04. Production and consumption has been since 2004 reduced to 15% of level from 1989 and it is to be phased out in 2015 according to Montreal Protocol.
References
- 1 2 3 4 5 "NIOSH Pocket Guide to Chemical Hazards #0197". National Institute for Occupational Safety and Health (NIOSH).
- 1 2 "Dichloromonofluoromethane". Immediately Dangerous to Life and Health. National Institute for Occupational Safety and Health (NIOSH).
External links
- International Chemical Safety Card 1106
- "NIOSH Pocket Guide to Chemical Hazards #0197". National Institute for Occupational Safety and Health (NIOSH).
- Termochemistry data at chemnet.ru
- Entry at Air Gas Liquide Encyclopaedia
This article is issued from Wikipedia - version of the 9/11/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.