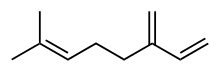
Myrcene
 | |
 | |
| Names | |
|---|---|
| IUPAC name
7-Methyl-3-methylene-1,6-octadiene | |
| Identifiers | |
| 123-35-3 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:17221 |
| ChEMBL | ChEMBL455491 |
| ChemSpider | 28993 |
| ECHA InfoCard | 100.004.203 |
| KEGG | C06074 |
| PubChem | 31253 |
| |
| |
| Properties | |
| C10H16 | |
| Molar mass | 136.24 g·mol−1 |
| Density | 0.794 g/cm3 |
| Melting point | < −10 °C (14 °F; 263 K) |
| Boiling point | 166 to 168 °C (331 to 334 °F; 439 to 441 K)[2] |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Myrcene, or β-myrcene, is an olefinic natural organic hydrocarbon. It is more precisely classified as a monoterpene. Monoterpenes are dimers of isoprenoid precursors, and myrcene is one of the most important. It is a component of the essential oil of several plants including bay, cannabis, ylang-ylang, wild thyme, parsley,[3] and hops.[4][5] It is produced mainly semi-synthetically from myrcia, from which it gets its name. It is a key intermediate in the production of several fragrances. α-Myrcene is the name for the structural isomer 2-methyl-6-methylene-1,7-octadiene, which is not found in nature and is little used.[6]
Biosynthesis and production
Terpenes arise naturally from dehydration of terpenol geraniol. It could in principle be extracted from any number of plants, for example wild thyme, the leaves of which contain up to 40% by weight of myrcene. The current route to commercial samples is by the pyrolysis (400 °C) of β-pinene, which is obtained from turpentine.[6]
Use in fragrance industry
Myrcene is an important intermediate used in the perfumery industry. It has a pleasant odor, but is rarely used directly. It is also unstable in air, tending to polymerize. Samples are stabilized by the addition of alkylphenols or tocopherol. It is thus more highly valued as an intermediate for the preparation of flavor and fragrance chemicals such as menthol, citral, citronellol, citronellal, geraniol, nerol, and linalool. Treatment of myrcene with hydrogen chloride gives geranyl chloride, neryl chloride, and linalyl chloride. Treatment of these compounds with acetic acid gives geranyl acetate, neryl acetate, and linalyl acetate, respectively. These esters are then hydrolyzed to the corresponding alcohols. Myrcene is also converted to myrcenol, another fragrance found in lavender, via uncatalyzed hydroamination of the 1,3-diene followed by hydrolysis and Pd-catalyzed removal of the amine.
As 1,3-dienes, both myrcene and mycenol undergo Diels-Alder reactions with several dienophiles such as acrolein to give cyclohexene derivatives that are also useful fragrances.[2]
Pharmacological effects
Myrcene has an analgesic effect and is likely to be responsible for the medicinal properties of lemon grass tea. It has antiinflammatory properties through Prostaglandin E2.[7] The analgesic action can be blocked by naloxone or yohimbine in mice which suggests mediation by "alpha 2-adrenoceptor stimulated release of endogenous opioids".[8]
Very high doses of myrcene potentiate barbiturate induced sleep in mice,[9] further supporting the suggestions of sedative activity. It is likely that this is responsible for the use of hops[10] and lemongrass[7] as sedatives. The boiling point of myrcene is near 166-168 degrees Celsius (330-334 degrees Fahrenheit) and it has analgesic, anti-inflammatory, antibiotic and antimutagenic properties.[11]
Partial list of the plants that contain myrcene
As mentioned above, many plants contain myrcene, sometimes in very large amounts.[6]
See also
References
- ↑ Merck Index, 11th Edition, 6243
- 1 2 Fahlbusch, K.-G.; Hammerschmidt, F.-J.; Panten, J.; Pickenhagen, W.; Schatkowski, D.; Bauer, K.; Garbe, D.; Surburg, H. (2002). "Flavors and Fragrances". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a11_141.
- ↑ Orav et al. (12/01/2003) "Composition of the essential oil of dill, celery, and parsley from Estonia", Estonian Academy of Sciences: Chemistry
- ↑ Chyau, C.-C.; Mau, J.-L.; Wu, C.-M. (1996). "Characteristics of the Steam-Distilled Oil and Carbon Dioxide Extract of Zanthoxylum simulans Fruits". Journal of Agricultural and Food Chemistry. 44 (4): 1096–1099. doi:10.1021/jf950577d.
- ↑ Tinseth, G. (January–February 1993). "Hop Aroma and Flavor". Brewing Techniques.
- 1 2 3 Behr, A.; Johnen, L. (2009). "Myrcene as a Natural Base Chemical in Sustainable Chemistry: A Critical Review". ChemSusChem. 2 (12): 1072–1095. doi:10.1002/cssc.200900186. PMID 20013989.
- 1 2 Lorenzetti, B. B.; Souza, G. R. E. P.; Sarti, S. L. J.; Santos Filho, D.; Ferreira, S. R. H. (1991). "Myrcene mimics the peripheral analgesic activity of lemongrass tea". Journal of Ethnopharmacology. 34 (1): 43–48. doi:10.1016/0378-8741(91)90187-I. PMID 1753786.
- ↑ Rao, V. S.; Menezes, A. M.; Viana, G. S. (1990). "Effect of myrcene on nociception in mice". The Journal of pharmacy and pharmacology. 42 (12): 877–878. doi:10.1111/j.2042-7158.1990.tb07046.x. PMID 1983154.
- ↑ Do Vale, T. G.; Couto Furtado, E. C.; Santos, J. G.; Viana, G. S. (2002). "Central effects of citral, myrcene and limonene, constituents of essential oil chemotypes from Lippia alba (Mill.) n.e. Brown". Phytomedicine : international journal of phytotherapy and phytopharmacology. 9 (8): 709–714. doi:10.1078/094471102321621304. PMID 12587690.
- ↑ Wichtl, Max, ed. (2004). Herbal Drugs and Phytopharmaceuticals. Third Edition. Medpharm.
- ↑ Skunk Pharm, Cannabinoid and terpene info