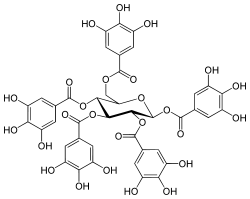
1,2,3,4,6-Pentagalloyl glucose
 | |
| Names | |
|---|---|
| IUPAC name
[(2S,3R,4S,5R,6R)-2,3,5-Tris[(3,4,5-trihydroxybenzoyl)oxy]-6-[(3,4,5-trihydroxybenzoyl)oxymethyl]oxan-4-yl] 3,4,5-trihydroxybenzoate | |
| Other names
1,2,3,4,6-Penta-O-galloyl-β-D-glucose 1,2,3,4,6-Pentakis-O-galloyl-beta-D-glucose beta-Penta-O-galloyl-glucose PGG 1,2,3,4,6-Penta-O-galloyl-beta-D-glucose | |
| Identifiers | |
| 14937-32-7 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:18082 |
| ChEMBL | ChEMBL382408 |
| ChemSpider | 58735 |
| ECHA InfoCard | 100.113.489 |
| PubChem | 65238 |
| |
| |
| Properties | |
| C41H32O26 | |
| Molar mass | 940.68 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
1,2,3,4,6-Pentagalloylglucose is the pentagallic acid ester of glucose. It is a gallotannin and the precursor of ellagitannins.[1]
Pentagalloyl glucose can precipitate proteins[2] including human salivary α-amylase.[3]
It may also be used in radioprotection.[4]
Natural occurrence
1,2,3,4,6-Pentagalloyl glucose can be found in Punica granatum (pomegranate),[5] Elaeocarpus sylvestris,[4] Rhus typhina (Staghorn sumac),[6] and Paeonia suffruticosa (Tree Peony).[7]
Biosynthesis
The enzyme beta-glucogallin-tetrakisgalloylglucose O-galloyltransferase uses 1-O-galloyl-beta-D-glucose and 1,2,3,6-tetrakis-O-galloyl-beta-D-glucose to produce D-glucose and pentagalloyl glucose.
Metabolism
Metabolism of ellagitannins
Tellimagrandin II is formed from pentagalloyl glucose by oxidative dehydrogenation and coupling of 2 galloyl groups.
β-glucogallin: 1,2,3,4,6-pentagalloyl-β-d-glucose galloyltransferase is an enzyme found in the leaves of Rhus typhina that catalyzes the galloylation of 1,2,3,4,6-penta-O-galloyl-β-D-glucose to 3-O-digalloyl-1,2,4,6-tetra-O-galloyl-β-d-glucose (hexa-galloylglucose).[6]
Chemistry
Pentagalloyl glucose can undergo oxidation reactions which are depending on the pH.[8]
References
- ↑ Cammann, Jessica; Denzel, Klaus; Schilling, Gerhard; Gross, Georg G. (1989). "Biosynthesis of gallotannins: β-Glucogallin-dependent formation of 1,2,3,4,6-pentagalloylglucose by enzymatic galloylation of 1,2,3,6-tetragalloylglucose". Archives of Biochemistry and Biophysics. 273 (1): 58–63. doi:10.1016/0003-9861(89)90161-6. PMID 2757399.
- ↑ Mechanisms of protein precipitation for two tannins, pentagalloyl glucose and epicatechin16 (4→8) catechin (procyanidin). Hagerman A. E., Rice M. E. and Ritchard N. T., Journal of agricultural and food chemistry, 1998, vol. 46, no7, pp. 2590-2595
- ↑ Evidence for pentagalloyl glucose binding to human salivary α-amylase through aromatic amino acid residues. Gyöngyi Gyémánt, Ágnes Zajácz, Bálint Bécsi, Chandran Ragunath, Narayanan Ramasubbu, Ferenc Erdődi, Gyula Batta and Lili Kandra, Biochimica et Biophysica Acta (BBA) - Proteins & Proteomics, Volume 1794, Issue 2, February 2009, pp. 291-296
- 1 2 Park, E; Lee, N. H.; Baik, J. S.; Jee, Y (2008). "Elaeocarpus sylvestris modulates gamma-ray-induced immunosuppression in mice: Implications in radioprotection". Phytotherapy research : PTR. 22 (8): 1046–51. doi:10.1002/ptr.2430. PMID 18570220.
- ↑ Tanaka, Takashi; Nonaka, Gen-Ichiro; Nishioka, Itsuo (1985). "Punicafolin, an ellagitannin from the leaves of Punica granatum". Phytochemistry. 24 (9): 2075. doi:10.1016/S0031-9422(00)83125-8.
- 1 2 Niemetz, Ruth; Gross, Georg G (1998). "Gallotannin biosynthesis: Purification of β-glucogallin: 1,2,3,4,6-pentagalloyl-β-d-glucose galloyltransferase from sumac leavesfn1fn1In honour of Professor G. H. Neil Towers' 75th birthday". Phytochemistry. 49 (2): 327. doi:10.1016/S0031-9422(98)00014-4.
- ↑ Fujiwara, H; Tabuchi, M; Yamaguchi, T; Iwasaki, K; Furukawa, K; Sekiguchi, K; Ikarashi, Y; Kudo, Y; Higuchi, M; Saido, T. C.; Maeda, S; Takashima, A; Hara, M; Yaegashi, N; Kase, Y; Arai, H (2009). "A traditional medicinal herb Paeonia suffruticosa and its active constituent 1,2,3,4,6-penta-O-galloyl-beta-D-glucopyranose have potent anti-aggregation effects on Alzheimer's amyloid beta proteins in vitro and in vivo". Journal of Neurochemistry. 109 (6): 1648–57. doi:10.1111/j.1471-4159.2009.06069.x. PMID 19457098.
- ↑ Reaction pH and protein affect the oxidation products of β-pentagalloyl glucose. Yumin C. and Hagerman A.E., Free radical research, 2005, vol. 39, no2, pp. 117-124